High-density cell cultivation is transforming the production of cultivated meat, making it more efficient and scalable. By growing animal cells in controlled environments, this method addresses the challenges of traditional farming, such as land use, water consumption, and greenhouse gas emissions. Key advantages include:
- Efficiency: High-density systems use bioreactors to grow cells in compact spaces, reducing resource waste.
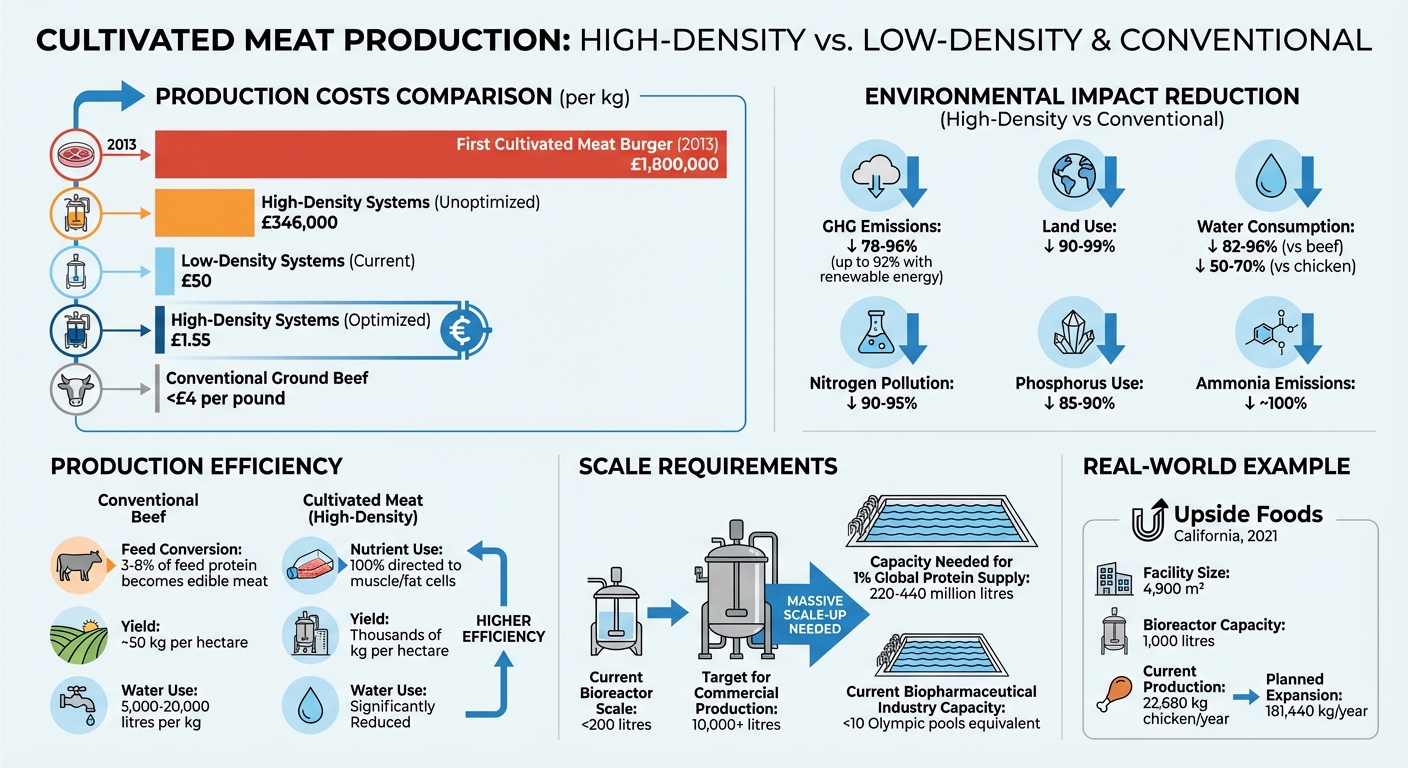
- Cost Reduction: Production costs can drop from £346,000/kg to as low as £1.55/kg with optimised methods.
- Environmental Impact: Cultivated meat can cut greenhouse gas emissions by up to 92% and land use by 90%.
- Nutritional Precision: These systems allow for tailored nutritional profiles, such as increased omega-3 content or reduced cholesterol.
Scaling remains a challenge, with current bioreactor capacities far below what's needed for global demand. However, advancements like perfusion systems, improved scaffolds, and cell line longevity are paving the way for large-scale production. Cultivated meat has the potential to meet rising protein needs while reducing environmental pressures and offering safer, antibiotic-free food options.
High-Density vs Low-Density Cultivated Meat Production: Cost, Environmental Impact & Efficiency Comparison
Cell growth modeling review for cultivated meat production: Recommendations to improve TEA models
sbb-itb-c323ed3
Why High-Density Cell Cultivation Is Necessary for Scaling Production
Scaling up Cultivated Meat production from lab experiments to large-scale food manufacturing is no small feat. Current biopharmaceutical methods typically operate at scales below 200 litres, which is far from sufficient for the demands of commercial food production. Mariana Petronela Hanga, a Lecturer in Cell Agriculture at University College London, explains:
"The highest scale that I am aware of is 200 litres. So there's definitely going to be a challenge there [reaching 10,000 litres]" [7].
To grasp the scale of this challenge, consider this: producing just 1 per cent of the global protein supply would require an estimated 220 to 440 million litres of fermentation capacity. That’s equivalent to 88 to 176 Olympic-size swimming pools. For context, the entire biopharmaceutical industry currently operates with fewer than ten such pools [7]. This gap highlights why high-density cell cultivation is essential - it’s the only way to make large-scale production feasible and efficient.
Problems with Low-Density Cultivation
Low-density cultivation methods simply don’t make sense for scaling. Take the example of the first Cultivated Meat burger, developed by Professor Mark Post of Maastricht University in 2013. It took two years to produce and cost a staggering £1.8 million per kilogram [6]. While costs have dropped significantly since then, low-density systems still hover around £50 per kilogram - a far cry from the price of conventional ground beef, which is typically less than £4 per pound [6] [7].
Cost isn’t the only issue. Low-density systems are resource-intensive, requiring large bioreactors and wasting expensive culture media. This inefficiency leads to higher water and energy use per kilogram of meat compared to optimised high-density systems [6] [8].
Another major drawback is quality. Low-density methods often produce loose cell aggregates or thin sheets, which lack the dense, fibrous texture of traditional meat. Achieving the familiar texture and "bite" of conventional beef or pork requires cells to grow in structured, high-density fibres - something that low-density systems struggle to achieve [6] [7].
Meeting Future Demand Through Scale
With global meat consumption projected to increase by 33.3 per cent by 2050 [6], addressing these inefficiencies is urgent. To meet even a fraction of this demand, Cultivated Meat production must scale up to industrial levels while remaining cost-effective and efficient.
High-density cultivation provides a promising path forward. When optimised, it could slash production costs from £346,000 per kilogram to as little as £1.55 per kilogram [6]. This dramatic reduction comes from better nutrient conversion, more efficient use of bioreactor space, and less reliance on costly culture media.
Some facilities are already paving the way. In November 2021, Upside Foods launched a 4,900‑square‑metre facility in Emeryville, California, equipped with 1,000‑litre bioreactors. This setup produces 22,680 kilograms of cultivated chicken annually, with plans to expand capacity to 181,440 kilograms. Such facilities are pilots for the high-density technology needed to reach production scales of millions of pounds [7].
Ultimately, high-density cultivation isn’t just about making Cultivated Meat viable - it’s about delivering on its environmental benefits. Without this efficiency, the promise of a more sustainable meat industry remains out of reach.
How High-Density Cell Cultivation Works
High-density cell cultivation tackles production challenges by leveraging advanced technology. At its core, this process relies on bioreactors - specialised vessels designed to create the ideal conditions for animal cells to grow and multiply efficiently.
Bioreactors and Controlled Growth Environments
Bioreactors serve as carefully engineered growth chambers, maintaining strict control over environmental factors. They ensure conditions such as a steady temperature of around 37°C and precise oxygen levels, both of which are critical to preventing nutrient shortages or cell damage. In the UK, the Food Standards Agency is overseeing a £1.6 million programme to ensure food produced in these environments meets safety standards for consumers [9].
Modern bioreactors are equipped with sensors and automated systems that monitor essential bioreactor metrics like cell density in real time. As cell populations grow, operators can adjust conditions to maintain balance. Mixing systems play a key role, ensuring all cells receive equal access to nutrients and oxygen, which prevents the formation of "dead zones" where cells might struggle to survive. This level of control is essential for managing cell nutrition using specially formulated culture media.
Culture Medium and Cell Nutrition
The culture medium is a nutrient-rich solution containing amino acids, vitamins, minerals, sugars, and growth factors. It must support densely packed cell populations while efficiently removing waste. In high-density systems, the medium's composition becomes even more critical. As cells grow closer together, they consume nutrients more rapidly and produce waste at an accelerated pace.
Maintaining the right balance is essential. If nutrients are insufficient, cell growth halts. Conversely, if waste products accumulate too quickly, cells experience stress or die. Achieving this balance is a key challenge in high-density cultivation.
Perfusion Systems for Continuous Nutrient Supply
Perfusion systems address the challenges of nutrient supply and waste removal by continuously refreshing the culture medium. Unlike batch systems, which add fresh medium at intervals, perfusion systems provide a constant flow of nutrients while simultaneously removing waste. This steady exchange allows cells to sustain high growth rates, even in extremely dense conditions.
These systems filter out waste products while keeping the cells inside the bioreactor. Fresh medium is supplied at a rate that matches the cells' consumption, creating a stable environment where cells can thrive. This continuous operation reduces downtime and enhances growth efficiency, making it a vital component for scaling up Cultivated Meat production to meet increasing demand.
Resource Efficiency and Environmental Benefits
High-density cell cultivation offers a direct route to feeding nutrients to cells, skipping the inefficiencies of animal metabolism. In traditional beef production, only 3–8% of the feed protein is converted into edible meat [10]. The rest supports non-edible functions like bone growth and other bodily structures. Cultivated Meat, on the other hand, focuses resources entirely on producing muscle and fat cells.
The potential for land savings is eye-opening. According to research from the University of Oxford, producing cultivated beef could use up to 99% less land compared to current livestock farming methods [5]. While conventional beef production yields about 50 kg per hectare, high-density systems can produce yields in the thousands [10]. Water use also sees dramatic reductions: cultivated systems consume 82–96% less water than conventional beef and 50–70% less than chicken [10]. To put it in perspective, producing 1 kg of conventional beef requires between 5,000 and 20,000 litres of water [10].
The environmental benefits of cultivated meat vs beef are another major advantage of high-density cultivation. When powered by renewable energy, these systems can slash greenhouse gas emissions by up to 92% compared to traditional beef production, with reductions ranging from 78–96% [10]. A significant contributor to this improvement is the elimination of methane emissions from enteric fermentation - a process in ruminants that generates a greenhouse gas 28 times more potent than CO₂ over 100 years [10].
Additionally, high-density systems dramatically cut nitrogen pollution by 90–95% and phosphorus use by 85–90% [10]. Unlike traditional livestock farming, which often leads to water pollution from animal waste runoff, the controlled environment of bioreactors avoids these issues altogether. Ammonia emissions, for instance, drop by nearly 100% due to the absence of animal waste [10]. These efficiencies also pave the way for better nutritional control within high-density cultivation systems.
Nutritional Control in High-Density Systems
High-density systems do more than just improve resource efficiency - they offer an impressive level of precision when it comes to nutritional control.
Adjusting Nutritional Composition
One of the standout features of high-density bioreactors is their ability to fine-tune the nutrients available to cultivated meat. Producers can carefully adjust levels of amino acids, glucose, fats, vitamins, and minerals to meet specific requirements [2][3]. This level of precision is simply not possible with traditional meat, where factors like genetics, feed, and environment heavily influence nutritional outcomes, compared to the precision of cultivated systems.
For instance, fat content can be modified to increase omega-3 levels or adjust intramuscular fat to meet specific goals. The medium can also be enriched with additional nutrients like vitamin A precursors, which enhances the overall nutritional value of the final product [2][3]. These tailored formulations open the door to creating meat that not only tastes good but also provides added health benefits.
Creating Healthier Meat Products
The ability to manage nutrients so precisely means cultivated meat can be designed with specific health benefits in mind. For example, high-density systems can produce meat with higher omega-3 content for heart health, lower cholesterol levels through controlled lipid inputs, and added vitamins that go beyond what’s typically found in conventional meat [1][5]. Dana Hunnes, PhD, MPH, RD from UCLA, highlights that nutrient content can even surpass that of traditional meat by adjusting the growth medium [3].
In addition to fortifying meat, researchers can adapt cell lines to eliminate allergens or introduce other desirable traits [2]. Some approaches even involve "exercising" muscle cells on scaffolds within bioreactors to boost protein levels while keeping fat and cholesterol in check [5]. Maintaining cell health in these dense environments is critical, and perfusion systems help by ensuring a steady supply of nutrients and closely monitoring metabolite levels [2][4]. This careful balance ensures consistent quality and nutritional value in the final product.
Current Challenges and Research Areas
High-density cultivation holds immense promise, but there are still technical obstacles and solutions to overcome before it can be scaled commercially. The sector has made substantial progress, with over $3.4 billion in investments by 2025 and a network of more than 140 companies spanning six continents. However, ongoing research remains essential [8].
Keeping Cells Healthy at High Density
One major challenge is maintaining cell health as densities increase. At approximately 3 × 10⁶ cells/ml, hydrodynamic forces and collisions begin to threaten cell attachment and viability [11]. Additionally, crowded conditions make it harder to ensure sufficient nutrient supply and efficient waste removal, demanding precise monitoring and control.
To address these issues, researchers are leveraging advanced tools like metabolic modelling and high-throughput automation. These methods help predict cell behaviour and refine media formulations. Upside Foods' EPIC facility in California provides a great example of this effort [7] [8]. As COO Amy Chen explains: "Large scale mammalian cell culture is well-established and has already successfully addressed many of the challenges... We have designed EPIC's equipment to allow us to simulate much larger cultivation vessels... to understand how cells are likely to react" [7]. The facility explores key factors like shear stress and gas exchange, aiming to maintain cell health at scale. These efforts tie directly into optimising physical support systems for dense cell growth.
Scaffold Design for Dense Cell Growth
Dense cell populations require scaffolds that are both robust and scalable. Microcarriers have emerged as a promising solution, offering high surface-to-volume ratios suitable for large-scale stirred-tank bioreactors [11]. For instance, researchers have successfully grown bovine satellite cells on microcarriers at concentrations of 10 to 80 cm²/ml, achieving up to tenfold expansion without compromising cell growth [11].
Recent developments have focused on making scaffolds more efficient and affordable. In 2024, Xin et al. introduced rice bran-gel composite scaffolds for fish cell growth, which increased the material's elastic modulus by 1.8 times while cutting costs to just 37% of pure gelatin systems [6]. Similarly, Su et al. used sorghum prolamin-based 3D porous scaffolds to guide porcine muscle and fat cells, resulting in a cultured pork product with a 22.9% protein content [6]. These plant-based materials not only reduce costs but also enhance compatibility and structural stability during high-density cultivation. Alongside scaffold advancements, ensuring cells remain stable over time is equally critical.
Cell Line Stability Over Time
Maintaining stable cell characteristics over long cultivation periods is another key challenge for scaling production. Researchers are working on continuous cell lines capable of indefinite proliferation, shifting away from finite primary cells [8]. This involves adapting cells through serial subculturing to select for stable traits, as well as employing genetic engineering to enhance productivity and genomic stability [8].
There has been notable progress in this area. In 2023, a team at Northwestern University slashed the cost of standard stem cell medium by 97%, making consistent cell growth far more affordable [8]. Additionally, since 2023, all approved cultivated meat products have been produced without the use of animal serum [8]. Governments in countries like Singapore, the United States, Brazil, and India are also stepping in, establishing cell line biorepositories to provide standardised starter materials. The industry is now tracking more than 90 cell lines globally [8].
Conclusion
High-density cell cultivation is the driving force behind turning Cultivated Meat from a lab experiment into a viable food system. By achieving industrial-scale cell growth - like Vow’s operation of a 20,000-litre food-grade bioreactor [8] - the industry is showing it can produce real protein while using fewer resources.
The advantages of these systems are hard to ignore. Cultivated Meat, especially when powered by renewable energy, has the potential to slash greenhouse gas emissions by up to 92% and reduce land use by as much as 90% [8]. Beyond environmental benefits, these systems improve food safety by removing the risk of pathogen exposure. Impressively, as of 2025, all approved Cultivated Meat products are produced without antibiotics [8], directly addressing the global issue of antimicrobial resistance.
"By decoupling meat production from live animal rearing, cultured meat has the potential to reduce zoonotic spillover risk, minimize antibiotic reliance, [and] lessen environmental burdens." - Ana Carolina Agne Ferreira Zão et al., Journal of Food Science [12]
The technological strides in high-density cultivation are tackling challenges like maintaining cell health at extreme densities, creating cost-effective scaffolds, and stabilising cell lines. Companies and collaborations are working tirelessly to address these hurdles. For instance, Believer Meats has teamed up with GEA to advance production methods [12], and Multus Biotechnology launched a food-grade basal media in early 2025 to support these efforts [12].
Stay updated on the latest developments and regulations by visiting Cultivated Meat Shop. With high-density cultivation evolving rapidly, the vision of feeding more people with fewer resources is no longer just a hope - it’s becoming a reality.
FAQs
What counts as “high-density” in Cultivated Meat production?
In the world of cultivated meat, "high-density" describes cell cultures where a significant number of cells grow efficiently within bioreactors. To achieve this, methods such as microcarriers or scaffolds are used. These tools ensure that nutrients, oxygen, and other essential elements are distributed evenly, even in the compact spaces of the bioreactor.
The aim is to maximise the number of cells while maintaining their health and uniform growth. This balance is critical for scaling up production and ensuring the process remains efficient and viable.
Why are perfusion bioreactors so important for scaling?
Perfusion bioreactors play a crucial role in scaling up the production of cultivated meat. They ensure a steady and even supply of oxygen and nutrients while efficiently removing waste, even in large-scale systems. This balance supports higher cell densities and promotes consistent growth.
By keeping conditions optimal, perfusion systems address the limitations of traditional methods when scaled up. This makes them essential for improving production efficiency, reducing costs, and paving the way for industrial-scale cultivated meat manufacturing.
What needs to change for cultivated meat to become affordable in supermarkets?
Achieving supermarket-level prices for cultivated meat hinges on economies of scale. This means developing larger, more efficient bioreactors and simplifying supply chains to cut costs. One of the biggest challenges is the cost of growth media, which can account for as much as 95% of production expenses. To tackle this, breakthroughs in growth factors, media production techniques, and cell line development are crucial. Pairing these with progress in automation and bioprocessing could help make cultivated meat a cost-effective choice for everyday shoppers.













